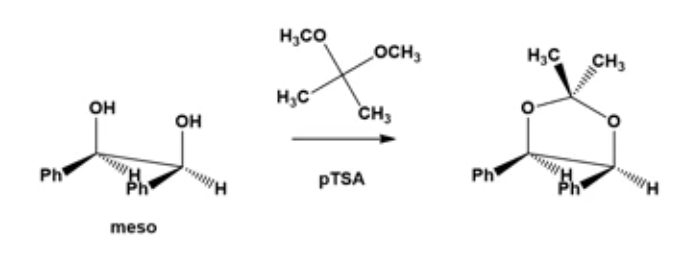 
SnCl 4 ( 6)) leads to a dramatic improvement in enantioselectivity in the allylboration of hydrocinnamaldehyde (93% ee).For example, the addition of cyclooctyl rings to the ortho and ortho' positions of the hydrobenzoin Notably, derivatives of hydrobenzoin in which the aromatic rings have been functionalized in the ortho and ortho' positions often display improved diastereo- or enantioselectivity over the parent diol 3. Moreover, the hydrobenzoin dimethyl ether 7 was shown to direct the asymmetric addition of organolithium reagents to arene tricarbonylchromium complexes and α,β-unsaturated aldimines. SnCl 4 complex 5 promotes the allylboration of hydrocinnamaldehyde with modest enantioselectivity (26% ee), and a hydrobenzoin–ytterbium complex was found to catalyze asymmetric aldol/Evans–Tishchenko reactions.For example, Hall reported that the hydrobenzoin Although hydrobenzoin (e.g., 3) has not been utilized to the same extent, it has also demonstrated utility as both a chiral ligand and auxiliary. In particular, C 2-symmetric diols, such as ( S)-BINOL ( 1) and (−)-TADDOL ( 2) ( Figure 1), have garnered considerable attention owing to the wide variety of asymmetric reactions promoted by these ligands and/or their derivatives. The discovery of new chiral ligands and auxiliaries continues to expand the frontiers of catalytic asymmetric synthesis.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
